
For a more detailed explanation with examples of Ramachandran plots, see Tutorial:Ramachandran Plot Inspection, Ramachandran Plot or Birkbeck's PPS95 course. observed) areas for non-glycine residues. If you plot phi against psi for each residue (so-called Ramachandran plot), you find that the phi/psi combination found in alpha helices fall into one of the three "allowed" (i.e. Īpart from the characteristic hydrogen bonding patterns, the other identifying feature of alpha helices are the main chain torsion angles. If you to a spacefilling representation, you can see how tightly packed the main chain is (no space in the middle). Because the amino acids connected by each hydrogen bond are four apart in the primary sequence, these main chain hydrogen bonds are called "n to n+4". The alpha helix is stabilized by (shown as dashed lines) from the of one amino acid to the of a second amino acid. (Stereo: ) In the following, the side chains are truncated at the beta carbon (green) to allow a better view of the main chain. The beta bend isĪn energy-effective method of accomplishing this goal.In an alpha helix, the main chain arranges in a with the pointing away from the helical axis. 
Reverse direc-tion many times in a typical globular protein. The third readily identified secondary structuralĮlement is the re-verse or beta bend (Fig. Oriented either parallel or antiparallel to the first. The amide hydrogens and the carboxyl groups areĭirected to either side and are available for hydrogen bonding to anotherīeta-strand lying alongside to form a beta sheet. The side chains of theĪmino acids are relatively unconstrained since alternate groups are directed From a top view the peptide backbone is relatively straight,īut in a side view the peptide backbone is pleated. In it the polypeptide chains are quite extended (Fig. The beta-strand is a second important structural element of proteins. 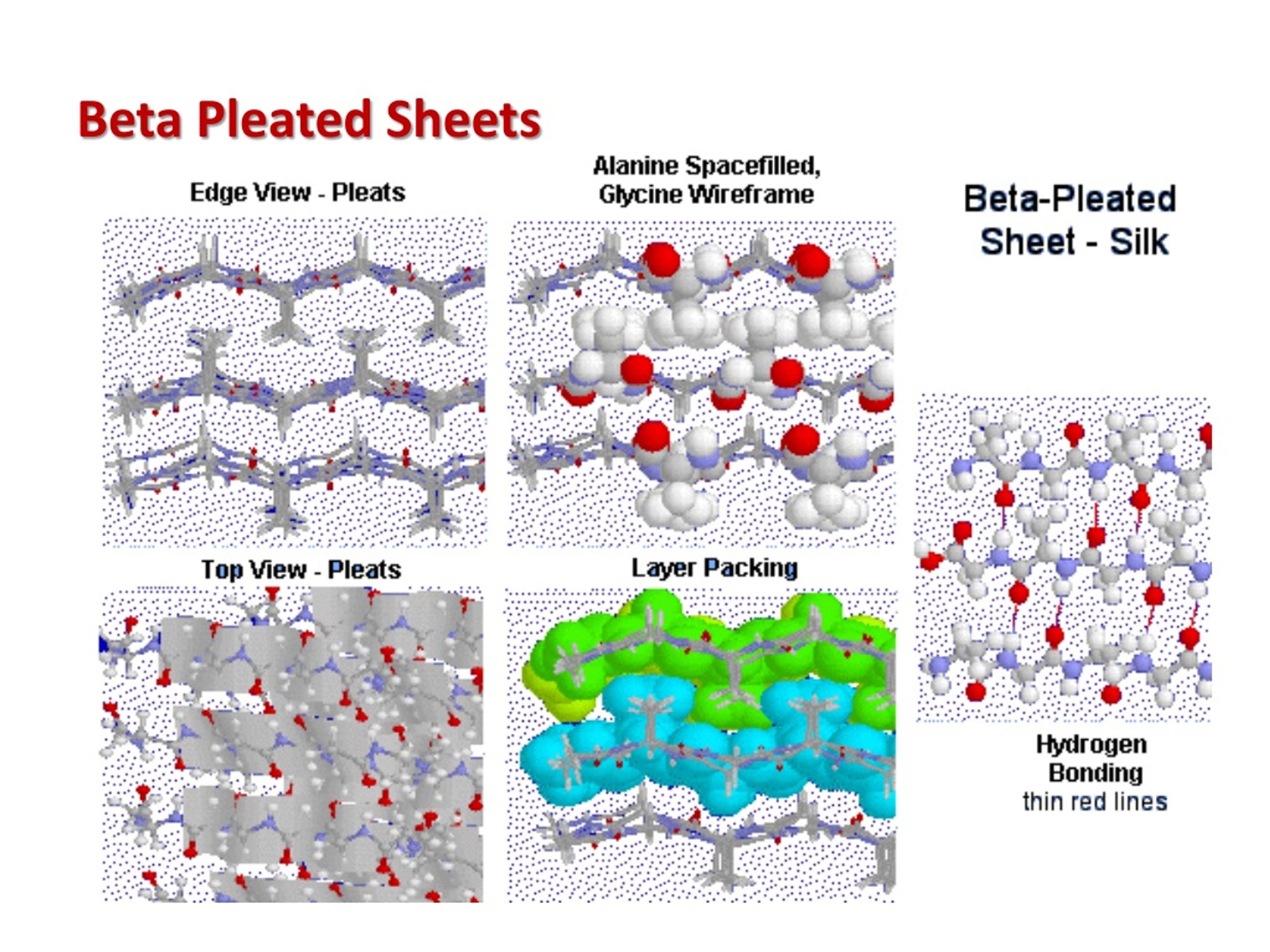
Hydrophobic amino acids along one face of alpha helices are frequently found inīundles containing two, three, or four alpha helices. They possess leucine residues seven amino acids apart. These activators are called leucine-zipper Proteins like myosin as well as in a class of transcrip-tional regulators thatĭimerize by these interactions. Occurs in structures called coiled coils. Third and then every fourth amino acid were hydrophobic, two such helices couldīind together through their parallel strips of hydrophobic amino acids. The fact that the alpha helix is nearly 3.5 amino acids per turn. Every third and then every fourthĪmino acid lies on one side of the helix (Fig. The amino acids winding around in a circle. If we look down the axis of an alpha helix, we see If they were precisely parallel to the axis, the helix pitch wouldīe 3.33 amino acids per turn, but due to steric constraints, the hydrogen bondsĪre somewhat skewed, and the average pitch is found to be 3.6 to 3.7 amino The side chains of the amino acids extend outwardįrom the helix, and the hydrogen bonds are nearly parallel to the helix axis Located three and a third amino acids away. 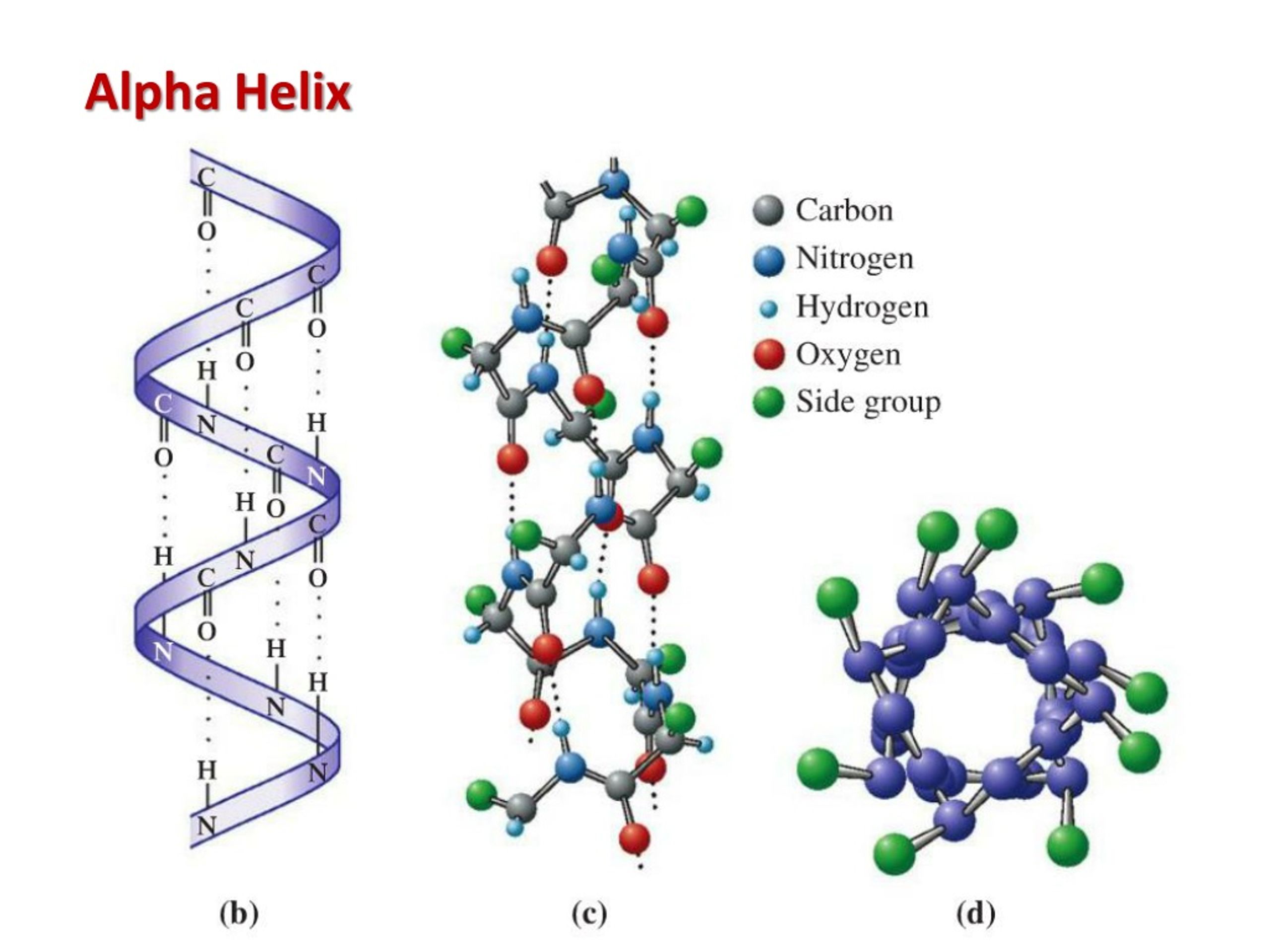
In the alpha helix, hydrogen bonds are formed between theĬarbonyl oxygen of one peptide bond and the amide hydrogen of the amino acid The alpha helix is found in most proteins and is a fundamental Even though the data were all there, it was This pre-diction came before identification of the alpha helix in X-rayĭiffraction patterns of proteins. Pauling and Cory from careful structural studies of amino acids and peptideīonds. The existence of the alpha helix was predicted by The Alpha Helix, Beta Sheet, and Beta Turn
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
